What happens to your medication before it reaches your hands? There’s one more step before the doctor, the pharmacy, and you. It begins much earlier, and it involves a long chain of manufacturers, storage facilities, transport networks, and distributors.
To be safe and efficient, that entire process needs a strong pharma supply chain. By “strong”, we mean something that keeps medicines stored at the right temperature, tracks their movement across borders, and prevents counterfeit products from entering legitimate channels.
When all of this is done the right way, hospitals and pharmacies get safe, high-quality medication on time.
But what’s behind the process and how does it work?
Source: Freepik
Why Supply Chain Strength Matters for Medication Safety
Before a medicine reaches your local pharmacy, it passes through several checkpoints. Each one is important for protecting quality and authenticity.
Here’s an example of the usual flow: a drug moves from a manufacturing facility to distribution centers, transport networks, wholesalers, and finally, to pharmacies or hospitals.
If just one step fails, the risk spreads throughout the entire system, and then patients might get contaminated batches, counterfeit drugs, or improperly stored products.
A report from Michigan State University’s A-CAPP Center talks about several risks that affect global pharmaceutical distribution. The 2019 Director’s Forum report explains that rogue online sellers and illegitimate imports create serious threats.
Such channels allow falsified or adulterated medicines to slip into legitimate markets and reach hospitals or patients.
The report also points to another issue: fragmented oversight between manufacturers, distributors, and pharmacies. When verification systems don’t align, gray-market diversions and unverified transactions become more likely.
Strong supply chains remove all these risks with strict verification and coordinated monitoring. Everyone does what they have to — manufacturers track production batches, distributors verify product origin, pharmacies confirm authenticity before dispensing medication.
It’s the only way for patients to receive safe, verified, and medication handled with care.
Temperature Control Protects Drug Quality
Medicines need strict temperature conditions from the very first moment they leave the manufacturer. Even a short exposure to heat or freezing temperatures can damage certain drugs.
Vaccines, insulin, biologic therapies, and many injectable medications all lose stability when storage conditions suddenly change. And a compromised product won’t just lose potency but could even become unsafe.
For this reason, temperature control is one of the most important safeguards in pharmaceutical logistics.
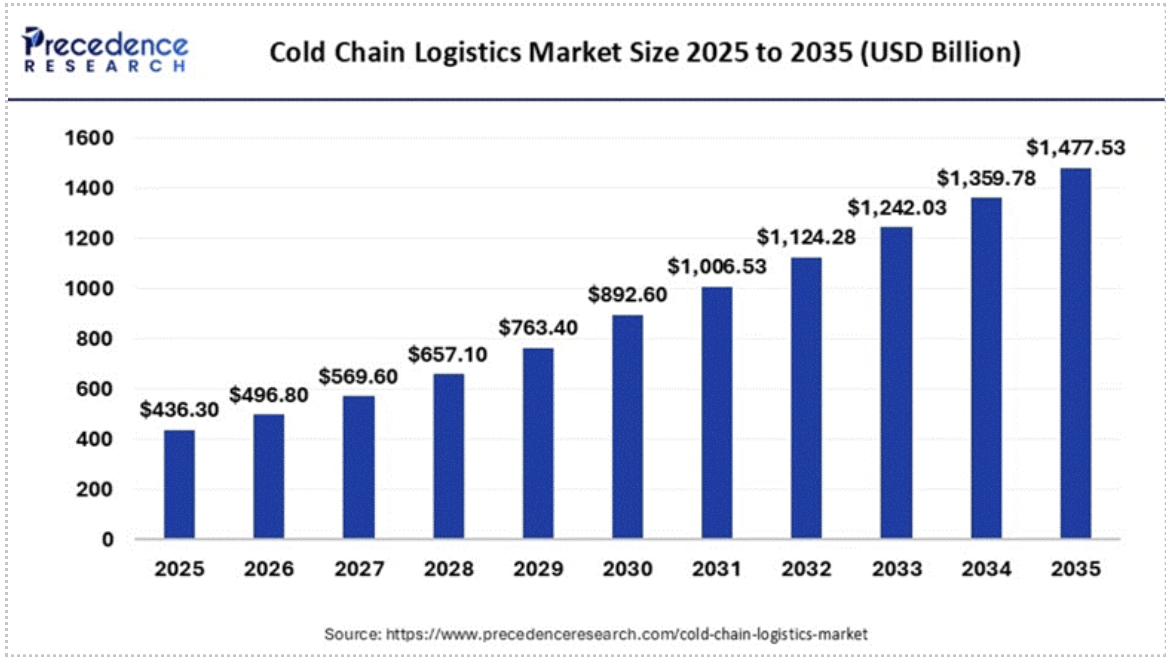
This requirement can also be seen in the major growth in cold-chain infrastructure. The global cold chain logistics market size reached $436.30 billion in 2025 and is expected to grow from $496.80 billion in 2026 to about $1,477.53 billion by 2035.
Source: precedenceresearch.com
So, the next time you need an mRNA vaccine, monoclonal antibodies, or any type of advanced biologic drug that’s temperature-sensitive, think about how narrow their temperature range is during all the transport.
To protect these products, supply chains often use these controlled systems:
- refrigerated and frozen transport vehicles
- temperature-monitored storage facilities
- insulated packaging designed for long transit periods
- digital sensors that record temperature throughout transport
Traceability Helps Detect Problems Early
Every package of medicine has information that allows it to be tracked across the supply chain. Manufacturers, distributors, and pharmacies need traceability to confirm that each product is legitimate and properly handled.
One modern solution is serialization, where every package gets a unique code that links it to a product batch and manufacturing location. When distributors scan that code at some point during transport, the system shows where the product traveled and who handled it along the way.
With such precise tracking comes quick verification, so pharmacies can confirm that the medicine came from an authorized distributor, and hospitals can verify that shipments match the correct production batch.
A lost package showing up where it shouldn’t be? The system can raise an alert.
Traceability also helps speed up recalls. If a manufacturer spots a problem with a specific batch, tracking data can help show where those units went. Rather than pulling large amounts from the market, companies can focus on the shipments that actually contain the affected products.
Reliable Distribution Keeps Medicines Available
Safe medication delivery needs more than just safety checks. Medicines also need to arrive where they are needed, when they are needed.
Hospitals, pharmacies, and clinics rely on steady distribution to keep treatments available for patients.
Pharmaceutical distribution networks need coordination when it comes to shipments for different regions and countries. For example, central warehouses often store large inventories, and regional ones supply pharmacies and hospitals based on local demand.
That way, medications move on time, and there’s no over- or understocking.
When there’s no coordination, there are problems. A single delay at a distribution hub can slow deliveries for the entire region.
Poor forecasting can empty shelves in one location and overstock another one. And since some medicines have short shelf lives, they can end up sitting unused and expiring.
Strong logistics systems usually prevent these situations with:
- demand forecasting based on prescription trends and seasonal needs
- regional warehouses that shorten delivery times
- verified distributor networks that follow regulatory standards
- controlled transport procedures that protect product quality
Thanks to all these elements working together, pharmacies get medicines on time, and patients don’t have to deal with unnecessary treatment delays.