HANGZHOU, China, April 3, 2026 /PRNewswire/ — A team led by principal investigators Bobo Dang and Ting Zhou at Westlake University/Westlake Laboratory reported in Science a high-throughput platform for engineering fast-acting covalent protein therapeutics. Their work, titled “A high-throughput selection system for fast-acting covalent protein drugs,” opens new avenues for next-generation biologics.
Covalent small-molecule drugs have shown great success in cancer therapy by forming irreversible bonds with their targets. This has inspired efforts to extend covalent strategies to protein therapeutics, especially engineered miniproteins. However, their development is limited by a kinetic mismatch: Miniproteins are rapidly cleared in vivo, whereas covalent bond formation is typically slow. In addition, high-throughput platforms for systematically optimizing covalent protein reactivity have been lacking.
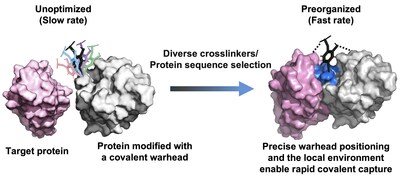
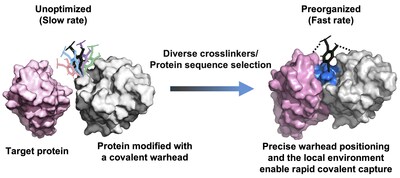
To address this challenge, the researchers proposed that precise spatial positioning of chemical warheads within protein scaffolds could enable molecular preorganization, thereby accelerating covalent bond formation without increasing intrinsic reactivity (Fig. 1).
Based on this concept, the team developed a high-throughput platform that combines yeast surface display with chemoselective protein modification to screen diverse crosslinkers and millions of protein variants. By optimizing warhead placement and the local chemical environment, the platform enables rapid and irreversible target engagement.
Using this platform, the researchers developed a covalent antagonist targeting PD-L1, termed IB101. Structural analysis revealed that IB101 forms a defined binding pocket that precisely positions the warhead in a reactive conformation, greatly accelerating covalent bond formation. Functionally, IB101 effectively blocks the PD-1/PD-L1 immune checkpoint pathway and demonstrates strong antitumor activity in mouse models. Notably, despite its short in vivo half-life, IB101 achieves durable target engagement and tumor suppression, outperforming conventional antibody-based therapies under comparable conditions.
The platform was further applied to cytokine engineering, leading to the development of a covalent IL-18 variant, IB201. This engineered cytokine rapidly forms a covalent interaction with its receptor, enhancing signaling strength and duration. In vivo studies showed that IB201 induces potent antitumor immune responses without detectable systemic toxicity. These results highlight the potential of covalent engineering to improve the efficacy and safety of cytokine-based therapies.
Beyond immunotherapy targets, the platform was also applied to develop a covalent inhibitor targeting the receptor-binding domain (RBD) of SARS-CoV-2. This molecule achieves durable viral neutralization, demonstrating the versatility of the approach across different therapeutic modalities.
This study establishes a general strategy for engineering fast-acting covalent protein therapeutics. By enabling covalent bond formation on timescales compatible with rapid in vivo clearance, the platform overcomes a fundamental limitation in the field.
These findings provide a new framework for designing biologics with both rapid kinetics and sustained target engagement, with broad implications for cancer immunotherapy, antiviral therapy, and beyond.
Media Contact:
Chi Zhang
[email protected]
+86-15659837873
View original content to download multimedia:https://www.prnewswire.com/news-releases/fast-acting-covalent-protein-drugs-from-a-new-high-throughput-platform-302733816.html
SOURCE Westlake University