LONDON, April 30, 2026 /PRNewswire/ — Serac Healthcare Limited (‘Serac Healthcare’ or ‘the Company’), a clinical radiopharmaceutical company developing an innovative molecular imaging agent, 99mTc-maraciclatide, and the Nuffield Department of Women’s & Reproductive Health at the University of Oxford announce the journal publication of Phase 2 data from the ‘Detecting Endometriosis expressed in Tegrins using teChneTium-99m’ (DETECT) imaging study. The paper, published open access online in The Lancet Obstetrics, Gynaecology & Women’s Health, highlights that 99mTc-maraciclatide has potential as:
- A novel, non-invasive diagnostic test, particularly for superficial peritoneal endometriosis (SPE), which cannot be reliably detected by conventional imaging techniques and currently requires laparoscopic surgery for definitive diagnosis
- A tool to enable disease monitoring, eliminating uncertainty around disease recurrence
- A marker of treatment response, empowering the development of novel therapies
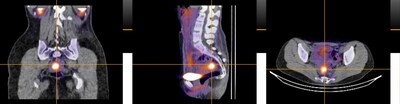
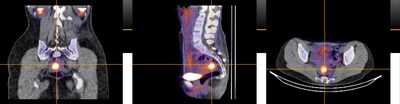
99mTc-maraciclatide is a gamma-emitting radiotracer that binds to αvβ3 integrin, which is upregulated during angiogenesis (new blood vessel formation), a cardinal feature of inflammatory diseases. The DETECT study describes, for the first time, the use of 99mTc-maraciclatide as a potential tool for the visualisation and diagnosis of endometriosis.
Key findings:
- The DETECT study demonstrated a high correlation between locations of maraciclatide uptake and laparoscopy across all types of endometriotic lesions, including superficial peritoneal endometriosis (SPE). SPE is the earliest stage of endometriosis, which is found in 80 per cent of all laparoscopically diagnosed disease
- Imaging with 99mTc-maraciclatide visualised endometriosis, detecting disease missed by conventional imaging methods
- Imaging results were concordant with the surgical presence or absence of endometriosis in 16/19 cases, with endometriosis imaged in 14/17 surgically positive participants, including two cases of thoracic endometriosis
- No false positives were reported in this study
- 99mTc-maraciclatide imaging was able to detect lesions across all endometriosis subtypes supporting the applicability of the scan to various patient groups
- 99mTc-maraciclatide was well tolerated and patient acceptability was high
David Hail, CEO of Serac Healthcare, commented:
“The completion and publication of this clinical study is a major milestone for Serac Healthcare. These data, from a representative patient population, including women receiving hormone therapy, provide evidence of maraciclatide’s anticipated real-world performance.
With FDA Fast Track Designation and agreed Phase 3 study designs, we are now advancing to validate these findings in larger trials and progress to regulatory submission.”
Dr Tatjana Gibbons, lead author on the paper and investigator on the study from the Nuffield Department of Women’s and Reproductive Health, University of Oxford, added,
“These exciting findings indicate that maraciclatide offers a highly promising diagnostic and monitoring tool, particularly for superficial peritoneal endometriosis, which is the most common and yet the hardest type of endometriosis to identify.
We are hugely grateful to the patients who have participated in the DETECT study without whom investigating this diagnostic approach would not have been possible.”
Professor Christian Becker, Co-Director of the Endometriosis CaRe Centre in Oxford, co-lead on the study and joint senior co-author on the paper, added:
“Novel, non-invasive diagnostic tests for endometriosis are a global research priority. The diagnostic challenge of endometriosis, which presents with varied and non-specific symptoms, is exacerbated by an absence of clinically validated biomarkers and the limitations of currently available imaging techniques. If these Phase II results are reproduced in the Phase 3 studies, maraciclatide has the potential to be an extremely valuable tool, as it could both reduce diagnostic delays and provide a validated endpoint for the development of new therapeutics.”
Professor Krina Zondervan, Co-Director of the Endometriosis CaRe Centre, Head of Department at the Nuffield Department of Women’s and Reproductive Health, Co-theme Lead for the NIHR Oxford Biomedical Research Centre’s Surgical Innovation Theme, co-lead on the study and joint senior co-author on the paper, said:
“Superficial peritoneal endometriosis, the most prevalent subtype of endometriosis, currently evades reliable detection, leaving women no choice for diagnosis other than invasive surgery. If these results are confirmed in larger Phase 3 studies, imaging with maraciclatide could transform clinical research and practice and potentially empower the development of treatments for women across the globe.”
About the Phase 2 trial:
This was an exploratory, open-label, single-centre, phase 2 study evaluating preoperative imaging in 20 individuals with suspected or confirmed pelvic or thoracic endometriosis using a SPECT-CT, with intravenous 99mTc-maraciclatide. The primary outcome was alignment of radiological and surgical findings in those patients completing both imaging and surgery. The surgical report on lesion type and location was compared to images for alignment. The study was funded by the NIHR Oxford Biomedical Research Centre and Serac Healthcare. Scans were performed at the Royal United Hospital, Bath.
Ten of the participants with SPE had prior imaging with traditional modalities (transvaginal (TV) ultrasound and/or MRI) in the last 12 months, none of which had detected SPE.
Phase 3 multi-centre international studies are planned to start later this year.
The open access paper is available here: https://www.thelancet.com/journals/lanogw/article/PIIS3050-5038(26)00048-8/fulltext
Further details on the study are available on ClinicalTrials.gov here: https://clinicaltrials.gov/study/NCT05623332?intr=NC100692&rank=4
Maraciclatide is for investigational use only and is not approved by the FDA or UK and European regulatory authorities.
Images are available on request.
Notes to Editors
Endometriosis
Endometriosis is a common inflammatory disease that affects up to one in 10 women of childbearing age, about 190 million women worldwide. Endometriosis occurs when tissue similar to the lining of the uterus is found outside the uterus, predominantly in the pelvis, but sometimes also elsewhere in the body, e.g. lungs. The presence of this ectopic endometrial tissue can lead to inflammation and distortion of normal anatomy, which can cause significant pain and infertility. Those with endometriosis typically experience an 8-12 year delay to diagnosis, requiring numerous visits to physicians and hospitals, along with multiple scans.
Photo: https://mma.prnewswire.com/media/2970155/DETECT_Study.jpg
Logo: https://mma.prnewswire.com/media/2970154/Nuffield_Oxford_Logo.jpg
Logo: https://mma.prnewswire.com/media/2613482/5947317/Serac_Healthcare_Logo.jpg
View original content to download multimedia:https://www.prnewswire.com/news-releases/endometriosis-imaging-study-published-in-the-lancet-obstetrics-gynaecology–womens-health-highlights-99mtc-maraciclatide-as-a-promising-diagnostic-and-monitoring-tool-302758770.html
SOURCE Serac Healthcare Limited