HENDERSON, Nev., March 31, 2026 /PRNewswire/ — Restore Robotics today announced it has received additional 510(k) clearances from the U.S. Food and Drug Administration (FDA) for the remanufacturing of two more da Vinci Xi® robotic surgical instruments. This milestone expands Restore Robotics’ growing portfolio of cleared instruments and further strengthens its position as a leader in robotic instrument remanufacturing.
With these latest clearances, Restore Robotics continues to deliver on its mission to provide hospitals and surgery centers with cost-effective, high-quality, and sustainable alternatives to purchasing new robotic instruments from original equipment manufacturers.
“These additional FDA clearances mark another major step forward in our effort to transform the economics of robotic surgery,” said Clif Parker, CEO of Restore Robotics. “By expanding our remanufacturing capabilities across more high-utilization instruments, we are enabling healthcare providers to significantly reduce costs while maintaining the performance and reliability surgeons expect.”
Through its established partnership with Encore Medical Device Repair, Restore Robotics will continue to scale distribution of its remanufactured instruments nationwide. The expanded product offering allows hospitals to capture greater savings across a broader portion of their robotic instrument spend.
Hospitals participating in the Restore Robotics remanufacturing program benefit from:
- Meaningful cost reductions versus OEM instruments
- Increased sustainability through instrument reuse
- Seamless integration into existing robotic surgery workflows
“Expanding the number of remanufactured robotic instruments available to hospitals is a natural progression in advancing responsible healthcare,” said Tom Milano, President and COO of Encore. “This continued collaboration represents a significant opportunity to improve both cost efficiency and environmental stewardship in surgical care.”
About Restore Robotics
Restore Robotics is a pioneer in the surgical robotics field, focused on the remanufacturing of robotic surgical instruments. With a state-of-the-art facility in Henderson, Nevada, the company delivers innovative, FDA-cleared solutions that enhance affordability and sustainability in robotic-assisted surgery.
About Encore Medical Device Repair
Encore Medical Device Repair was founded on the principle that medical device reuse should play a central role in responsible healthcare. By focusing on high-value, high-utilization instruments, Encore delivers industry-leading service, quality, and cost savings to healthcare providers.
Media Contact:
Clif Parker
CEO, Restore Robotics
[email protected]
678-619-0011
www.restorerobotics.com
Disclaimer:
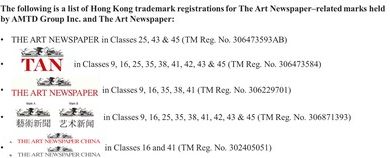
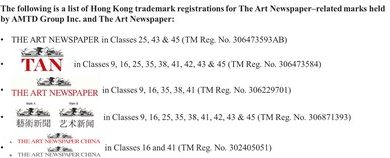
Intuitive Surgical, Inc. has granted approval under its contracts for Restore Robotics to remanufacture any EndoWrist that falls under the scope of any 510(k) clearance from the FDA. EndoWrist®, da Vinci®, da Vinci Xi®, and da Vinci Si® are registered trademarks of Intuitive Surgical, Inc. Restore Robotics is an independent entity and is not affiliated with Intuitive Surgical, Inc. or its affiliates. All registered trademarks and product names are the property of their respective owners.
View original content to download multimedia:https://www.prnewswire.com/news-releases/restore-robotics-receives-fda-510k-clearance-for-two-additional-da-vinci-xi-robotic-instruments-302729165.html
SOURCE Restore Robotics